Human Serum Albumin, Excipient, USP/EP - NOVA Biologics, Inc.
Human Serum Albumin, Excipient, USP/EP - NOVA Biologics, Inc.
NOVA is the authorized distributor of Human Serum Albumin, USP/EP manufactured by Octapharma to the global market. Our HSA is manufactured per US and European Pharmacopeia specifications in Octapharma’s FDA, EMA, and GMP certified facilities using only US sourced human plasma. This HSA is acceptable for use as an excipient and/or for further manufacture of

Oral abstracts (OAS) - 2023 - Allergy - Wiley Online Library

WO2009073569A2 - Protein formulations and methods of making same

Human Serum Albumin (HSA) Diagnostic - Lyophilized - NOVA

US9085619B2 - Anti-TNF antibody formulations - Google Patents

Don't forget to take care of your hearts on World Heart Day
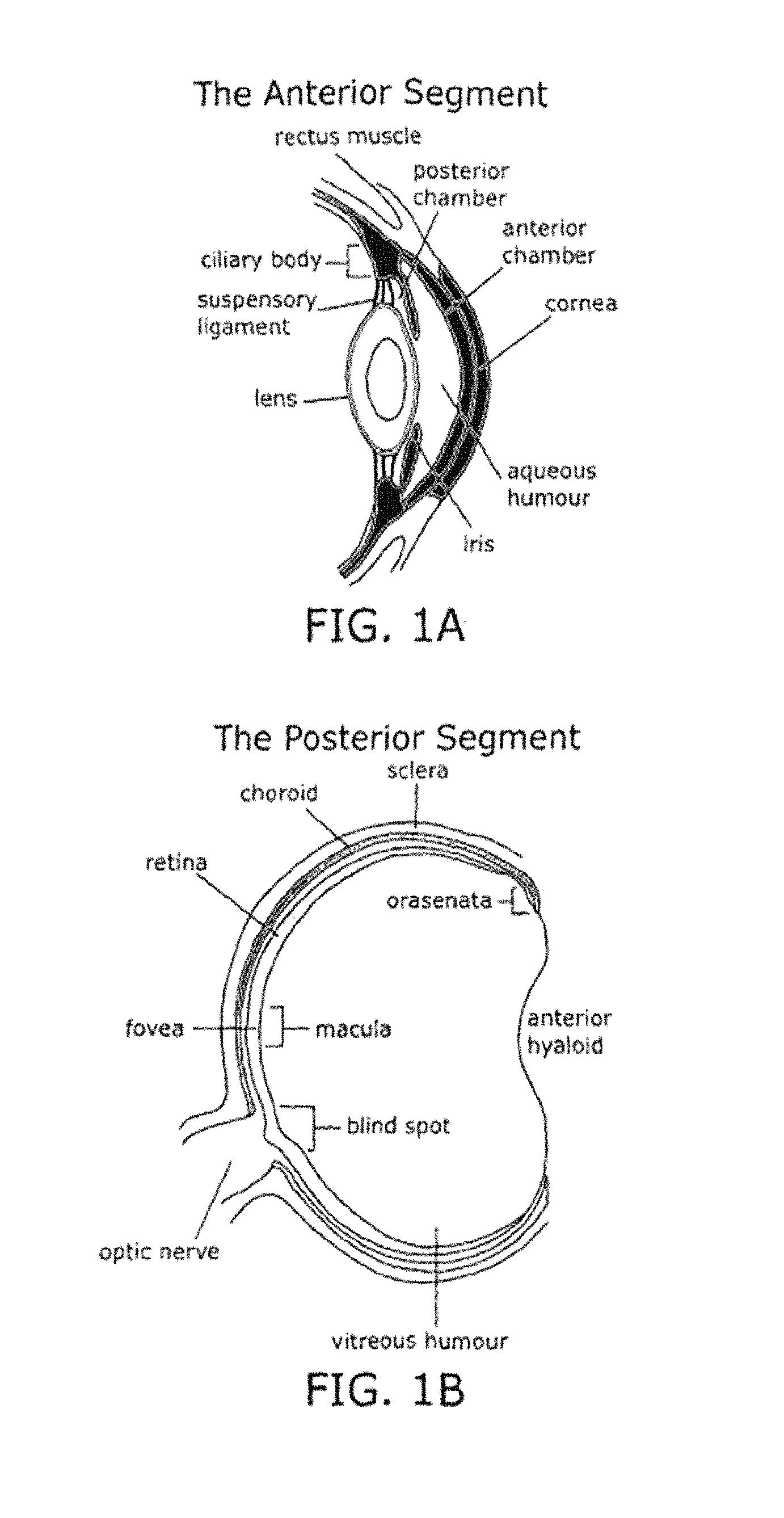
Methods of selecting compstatin mimetics Patent Grant Deschatelets

Setting Biological Process Specifications

Prefilled Syringes & Injection Devices - ONdrugDelivery - Issue 61

Setting Biological Process Specifications
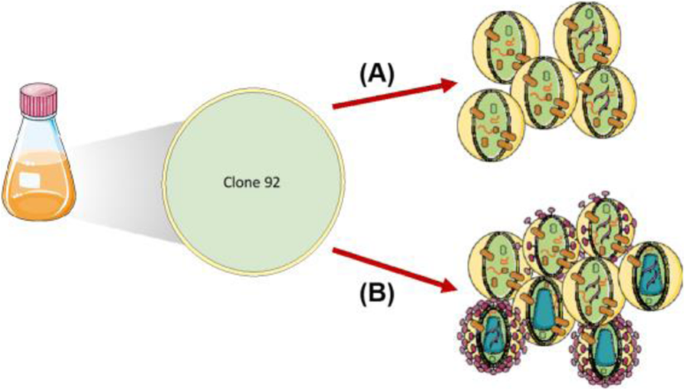
Abstracts from the 26th European Society for Animal Cell

Solvents Product and Supplier Directory - CPHI Online

PDF) Enhancement of The Stability of Human Growth Hormone by Using

Excipient Grade (Recombinant Human Serum Albumin)-Oryzogen

Albumin, Human

NOVA Biologics, Inc. on LinkedIn: Did you know? “BovoStar” Premium


